“Some of my patients couldn’t tolerate oral finasteride. A targeted topical technology didn’t exist yet, so we built it.”
Topical Finasteride with SiloxysSystem™ Gel
- Patented gel designed to minimize side effects
- Trusted by over 100+ hair restoration clinics worldwide
- Created by leading hair loss doctors
Individual response to medication varies.
"2.5% sounds high — won't that increase my risk of side effects?"
SiloxysSystem gel keeps medication anchored at the scalp. Because the gel holds the medication tightly, a higher concentration is needed to allow medication to move from the gel into the scalp. That’s why dosages can’t be directly compared to pills or other formulas. What matters most is how much reaches your scalp and how little spreads elsewhere, not just the percentage on the label.
This is a compounded prescription medication. It is not FDA-approved. The FDA does not verify the safety or effectiveness of compounded medications. Compounded medications may be prescribed off-label by a licensed physician for patients for whom the potential benefits outweigh the risks. Your prescribing physician will review your full medical history and determine if this medicine is appropriate for you.
Why SiloxysSystem™ works differently
-
01
Designed to release finasteride gradually, not all at once
-
02
92% less absorbed into the blood than pills
-
03
Limits body absorption better than tested liquid topicals
Results from our patients


“Seeing my hair stabilize and rebound, especially around my hairline, was exactly what I was hoping for.”
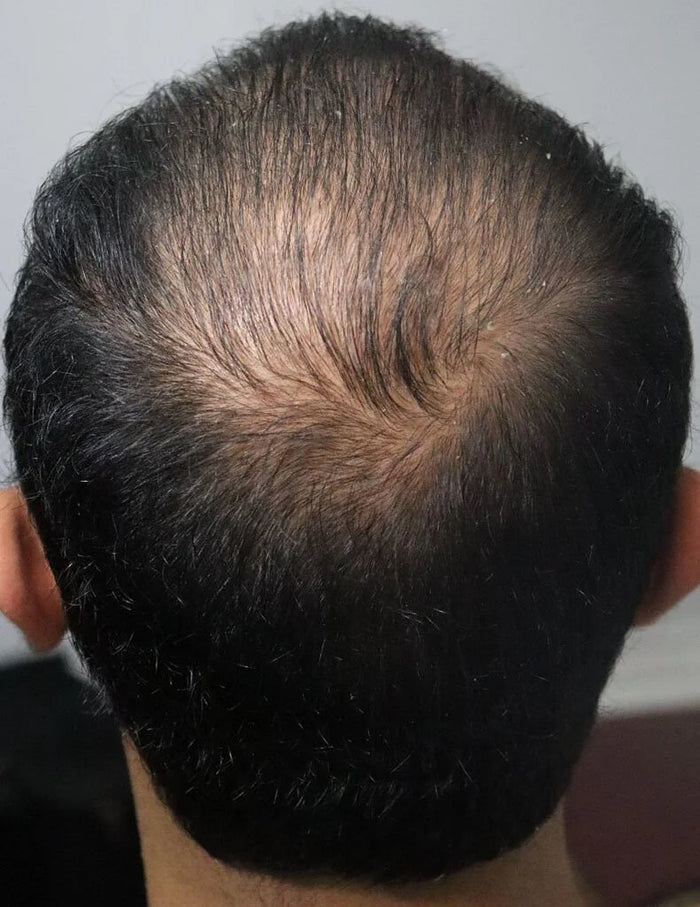
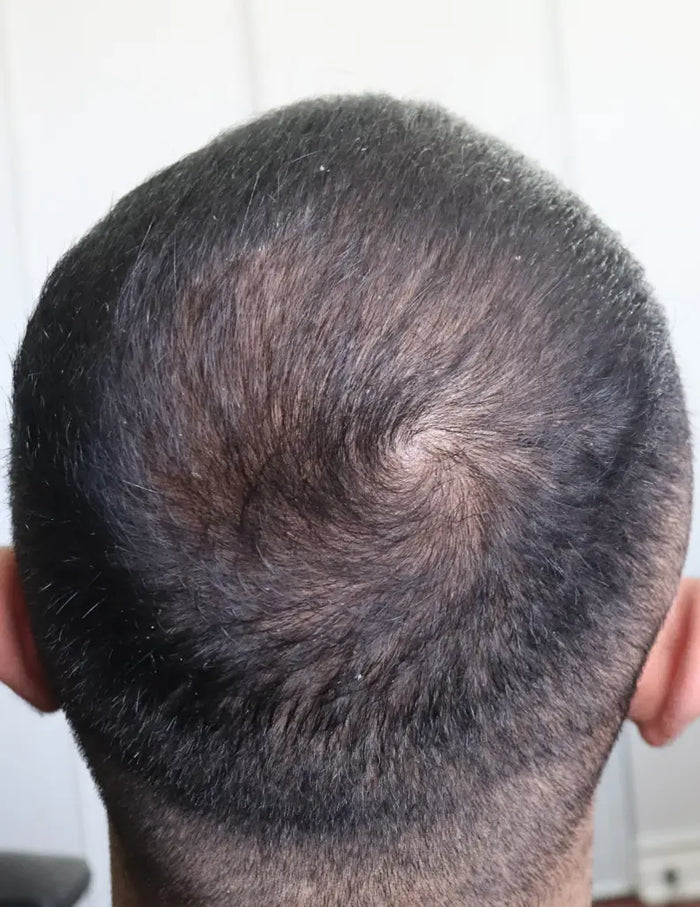
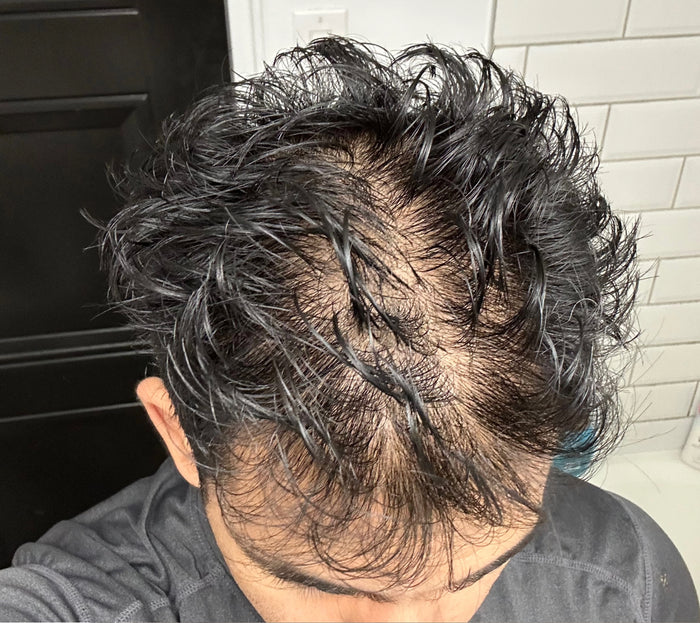
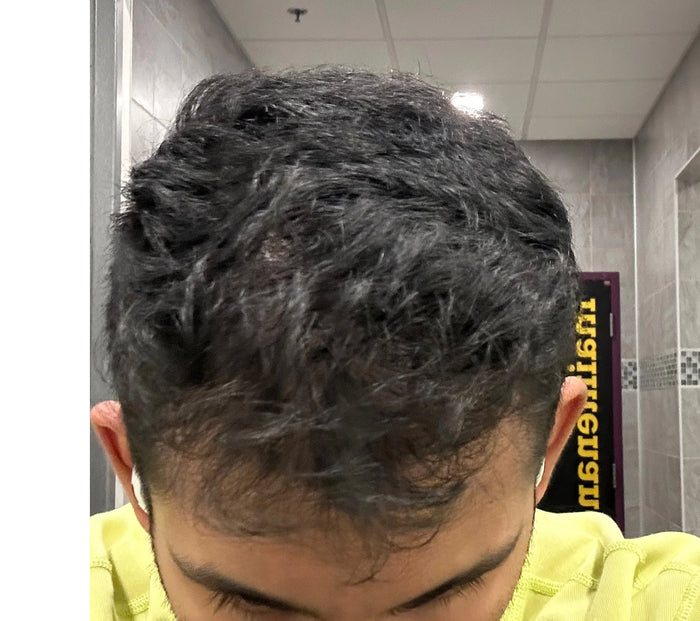
“XYON's Topical Finasteride helped me reverse my hair loss and increase my hair thickness and it's been the game changer I always needed to stop my hair loss.”


“I saw improvements everywhere. It’s difficult to gauge because of my hair transplants, but I know the finasteride from XYON helped my hair, especially the miniaturized hair.”
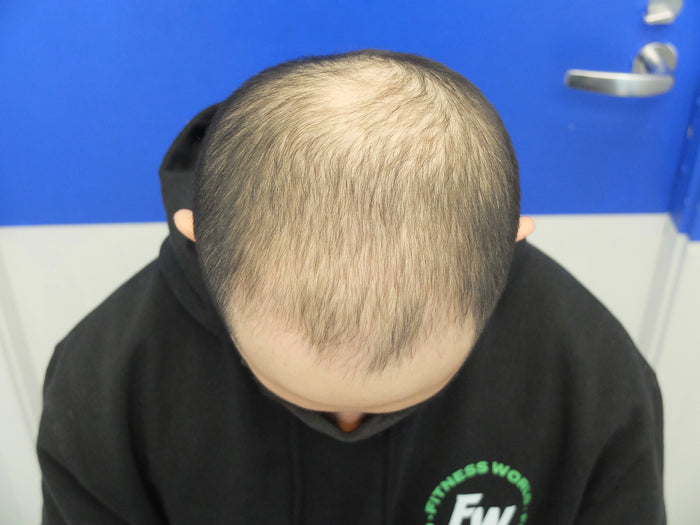
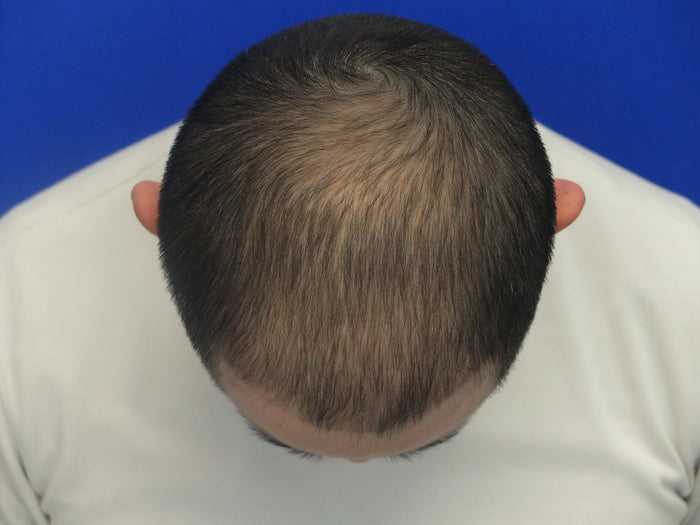
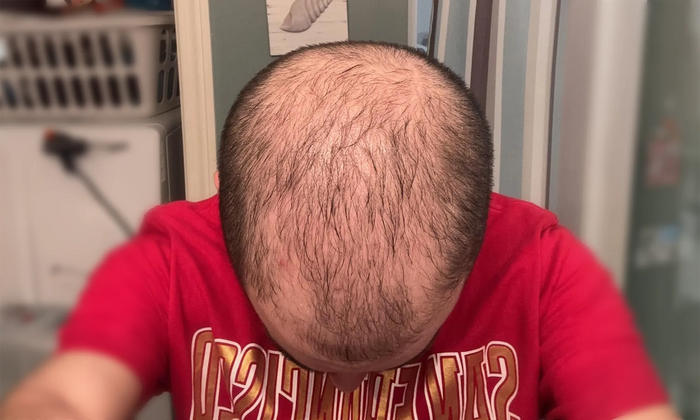
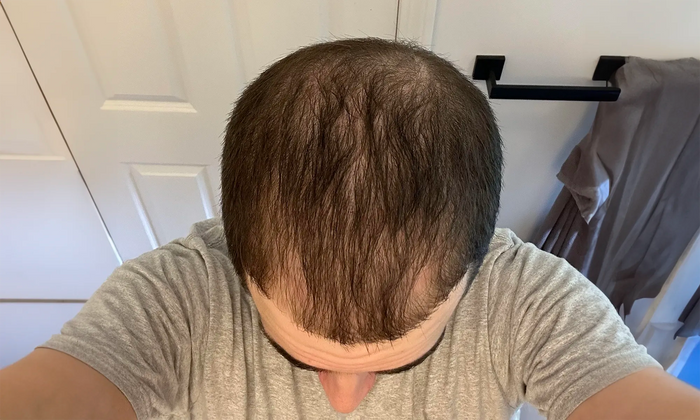
“I've seen incredible results with finasteride and minoxidil after 4 months…”
Individual results may vary. The case studies and testimonials shown reflect the personal experiences and opinions of the individuals featured. They are not statements or claims by XYON Health and should not be interpreted as guarantees of outcome. Results depend on factors including genetics, adherence, and underlying medical conditions. Always consult a qualified healthcare provider before starting any treatment.
How to apply SiloxysSystem™ Gel
Step 1 — Unlock and press
Turn the safety lock to open. Press the pump once to dispense one full dose (1mL) onto your fingertips — it may take a few presses the first time to prime the pump — and get a full dose. One dose covers most areas of thinning. If you're treating a larger area, follow your prescriber's dosing instructions.
Step 2 — Apply to scalp
Separate your hair to expose the affected area of scalp. Distribute small amounts of gel across the affected areas, then massage gently for 20–30 seconds.
Step 3 — Style as usual
Wash hands with soap and warm water after applying. Style as usual — try not to disturb the gel. It works best when it stays in contact with your scalp for 4–8 hours after application.

How XYON works

Complete your consultation
Share your medical history and select your treatment preference. Upload photos of your hair. Your doctor may ask you to verify your identity through your secure account.
Connect with a doctor
A doctor reviews your consultation and decides on the ideal treatment plan for your needs.
Start your prescription
Your medication is fulfilled and shipped by the pharmacy. Most orders ship within one week of fulfillment. — we'll send tracking details once your prescription ships.
Ongoing support
Log in anytime to request changes to your plan, medication, or dosage. Changes to your medication or dosage require doctor approval. Have questions? We're here.
Created in the clinic, not the boardroom.
Founded by a world-renowned hair surgeon and a life sciences CEO with 25 years in drug development.
“Our innovation rethought how medication is delivered to the scalp, giving patients worried about side effects another option.”
Further reading
View all hair loss resources →




XYON Topical Finasteride customer reviews
Individual experiences vary. Customer reviews reflect personal opinions and outcomes, not guarantees of results. Results depend on factors including genetics, adherence, and underlying medical conditions. Always consult a qualified healthcare provider before starting any treatment. Reviews are collected through Yotpo, our independent review provider.

